Production QA QC Microbiology Engineering Jobs at CEPH Lifesciences roles at its Baddi pharma facility.
Job Type: Full-Time
Industry: Pharmaceutical Manufacturing
Experience Required: 1–13 Years
Location: Baddi, Himachal Pradesh
Application Mode: Walk-In Interview / Email Support
Job Status: Verified Job
About the Company
CEPH Lifesciences is a fast-growing pharmaceutical manufacturing company associated with Nectar Lifesciences, one of India’s established names in the pharma sector. With a strong focus on quality, compliance, and continuous improvement, CEPH Lifesciences is steadily expanding its footprint in regulated and semi-regulated markets. Production QA QC Microbiology Engineering Jobs
The company operates a modern manufacturing facility located at Village Bhatoli Kalan, Adjoining Jhar Majri, Baddi, Himachal Pradesh. This facility is equipped with advanced infrastructure for injectable, oral solid dosage, quality control, microbiology, and engineering operations. CEPH Lifesciences is committed to delivering high-quality pharmaceutical products while maintaining strict adherence to GMP, regulatory guidelines, and ethical manufacturing practices.
CEPH Lifesciences offers professionals a stable work environment, opportunities for long-term growth, and exposure to diverse pharmaceutical operations across multiple departments.
Job Details
- Hiring Departments: Production, Quality Assurance (QA), Quality Control (QC), Microbiology, Engineering
- Company: CEPH Lifesciences
- Employment Type: Full-Time
- Work Location: Baddi, Himachal Pradesh
- Experience Range: 1 to 13 years
- Educational Qualifications: 10+2 / ITI, B.Pharma, M.Pharma, M.Sc, B.Tech (as applicable)
- Interview Type: Walk-In Interviews (Ongoing)
This hiring drive is suitable for both junior-level and experienced pharma professionals looking to grow in manufacturing, quality, microbiology, or engineering functions. Production QA QC Microbiology Engineering Jobs
Job Description
CEPH Lifesciences is conducting ongoing walk-in interviews to strengthen its Production, QA, QC, Microbiology, and Engineering teams. Selected candidates will be responsible for executing day-to-day pharmaceutical operations, ensuring compliance with GMP standards, maintaining documentation, and supporting continuous improvement initiatives.
The roles involve hands-on work in injectable and oral dosage manufacturing, quality systems, analytical testing, microbiological analysis, and maintenance of critical utilities and engineering systems. Professionals joining CEPH Lifesciences will contribute directly to the production of high-quality pharmaceutical products in a regulated manufacturing environment.
Skills / Qualifications
Educational Qualifications
Candidates must possess relevant qualifications based on the role applied for: Production QA QC Microbiology Engineering Jobs
- 10+2 or ITI for operator and technician roles
- B.Pharma or M.Pharma for officer, executive, and QA/QMS roles
- M.Sc or B.Tech (Biotechnology/Microbiology) for microbiology roles
- ITI or B.Tech for engineering and utility roles
Experience Requirements
- Minimum 1 year of experience for junior roles
- Up to 13 years of experience for senior and managerial roles
- Prior experience in pharmaceutical manufacturing is preferred
Key Skills
- Strong understanding of GMP, SOPs, and regulatory compliance
- Hands-on experience with pharma equipment and processes
- Good documentation and audit readiness skills
- Team collaboration and problem-solving abilities
- Willingness to work in a plant-based manufacturing environment
Production QA QC Microbiology Engineering Jobs
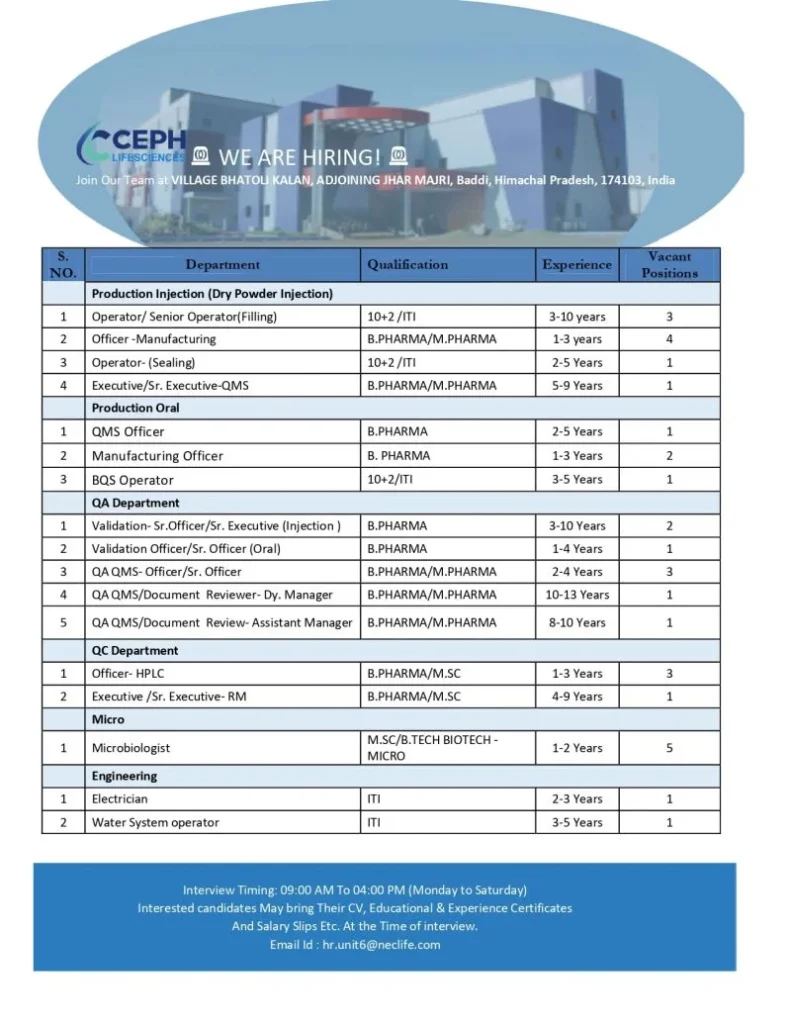
Open Positions and Role Details
Production – Injection (Dry Powder Injection)
- Operator / Senior Operator (Filling):
Qualification: 10+2 / ITI
Experience: 3–10 years
Vacancies: 3 - Officer – Manufacturing:
Qualification: B.Pharma / M.Pharma
Experience: 1–3 years
Vacancies: 4 - Operator (Sealing):
Qualification: 10+2 / ITI
Experience: 2–5 years
Vacancies: 1 - Executive / Senior Executive – QMS:
Qualification: B.Pharma / M.Pharma
Experience: 5–9 years
Vacancies: 1
Production – Oral
- QMS Officer:
Qualification: B.Pharma
Experience: 2–5 years
Vacancies: 1 - Manufacturing Officer:
Qualification: B.Pharma
Experience: 1–3 years
Vacancies: 2 - BQS Operator:
Qualification: 10+2 / ITI
Experience: 3–5 years
Vacancies: 1
Production QA QC Microbiology Engineering Jobs
Quality Assurance (QA) Department
- Validation Sr. Officer / Sr. Executive (Injection):
Qualification: B.Pharma
Experience: 3–10 years
Vacancies: 2 - Validation Officer / Sr. Officer (Oral):
Qualification: B.Pharma
Experience: 1–4 years
Vacancies: 1 - QA / QMS Officer / Sr. Officer:
Qualification: B.Pharma / M.Pharma
Experience: 2–4 years
Vacancies: 3 - QA QMS / Document Reviewer – Dy. Manager:
Qualification: B.Pharma / M.Pharma
Experience: 10–13 years
Vacancies: 1 - QA QMS / Document Reviewer – Assistant Manager:
Qualification: B.Pharma / M.Pharma
Experience: 8–10 years
Vacancies: 1
Production QA QC Microbiology Engineering Jobs
Quality Control (QC) Department
- Officer – HPLC:
Qualification: B.Pharma / M.Sc
Experience: 1–3 years
Vacancies: 3 - Executive / Sr. Executive – RM:
Qualification: B.Pharma / M.Sc
Experience: 4–9 years
Vacancies: 1
Microbiology Department
- Microbiologist:
Qualification: M.Sc / B.Tech (Biotechnology – Microbiology)
Experience: 1–2 years
Vacancies: 5
Engineering Department
- Electrician:
Qualification: ITI
Experience: 2–3 years
Vacancies: 1 - Water System Operator:
Qualification: ITI
Experience: 3–5 years
Vacancies: 1
Key Responsibilities (General)
- Operate and monitor pharmaceutical manufacturing equipment
- Ensure strict compliance with GMP, SOPs, and safety standards
- Perform in-process checks, validation, and documentation
- Conduct analytical and microbiological testing as applicable
- Handle QMS activities, audits, and document review
- Maintain engineering utilities such as water systems and electrical equipment
- Collaborate with cross-functional teams to ensure product quality and efficiency
Production QA QC Microbiology Engineering Jobs
Salary & Benefits / Perks
CEPH Lifesciences offers competitive salary packages based on role, experience, and skill set. Production QA QC Microbiology Engineering Jobs
- Junior-level roles: Approximately ₹2.5 – ₹8 LPA
- Senior and managerial roles: Up to ₹10 – ₹18 LPA
Additional benefits may include:
- Performance-based incentives
- Health insurance coverage
- Provident Fund (PF) and ESI
- Stable employment in a growing pharma organization
- Career growth and internal advancement opportunities
Production QA QC Microbiology Engineering Jobs
Why You Should Join CEPH Lifesciences
CEPH Lifesciences provides an excellent opportunity for pharma professionals to build a long-term career in a growing manufacturing setup. The company’s focus on quality, compliance, and employee development makes it an attractive workplace for those seeking stability and growth. Production QA QC Microbiology Engineering Jobs
Working at the Baddi facility offers exposure to injectable and oral manufacturing, quality systems, microbiology, and engineering operations under regulated conditions. CEPH Lifesciences values dedication, technical competence, and continuous improvement, making it an ideal environment for career progression.
How to Apply
This hiring is through walk-in interviews, and no prior application is required. Production QA QC Microbiology Engineering Jobs
Walk-In Interview Details
- Interview Days: Monday to Saturday
- Timing: 09:00 AM to 04:00 PM
- Venue:
CEPH Lifesciences Facility
Village Bhatoli Kalan,
Adjoining Jhar Majri,
Baddi, Himachal Pradesh – 174103
Documents to Carry
- Updated resume
- Educational certificates
- Experience certificates
- Salary slips (if applicable)
Contact for Queries
- Email: hr.unit6@neclife.com
FAQs
Who can apply for these roles?
Candidates with 1–13 years of experience in pharmaceutical manufacturing, quality, microbiology, or engineering can apply.
Are freshers eligible?
Some junior roles may consider candidates with minimal experience, but prior pharma exposure is preferred.
Is this a walk-in interview?
Yes, interviews are conducted daily from Monday to Saturday during working hours.
What is the job location?
All positions are based at CEPH Lifesciences, Baddi, Himachal Pradesh.
Is prior pharma experience mandatory?
Yes, prior pharmaceutical industry experience is preferred for most roles.
This is a strong opportunity for pharma professionals to join CEPH Lifesciences and grow their careers in a regulated, quality-driven manufacturing environment. Production QA QC Microbiology Engineering Jobs
For verified pharma job updates, visit PharmaJobHub.in – India’s trusted pharma career portal.
Note: This job information is sourced from publicly available official sources. We share it for reference purposes only and do not claim ownership of the job posting.



