Freshers Hiring for Production QA QC & Engineering Job at Sanolet Lifecare Ahmedabad with excellent career growth opportunities.
Introduction
Sanolet Lifecare Pvt. Ltd. is inviting applications from fresh graduates and experienced professionals for multiple roles in Production, Quality Assurance, Quality Control, and Engineering. This opportunity is ideal for candidates looking to build or advance their career in the pharmaceutical manufacturing sector. With positions open for both freshers and candidates having up to six years of experience, this hiring drive offers a strong platform to work in a regulated and growth-oriented environment.
Company Overview
Sanolet Lifecare Pvt. Ltd. is a WHO-GMP certified pharmaceutical manufacturing company based in Ahmedabad, Gujarat. The company is known for its specialization in dry powder injection formulations and follows stringent quality and regulatory standards. With a focus on innovation, compliance, and product quality, Sanolet Lifecare continues to expand its presence in the pharmaceutical industry while maintaining high operational excellence.
Job Overview
| Job Title | Production / QA / QC / Engineering Roles |
|---|---|
| Company Name | Sanolet Lifecare Pvt. Ltd. |
| Location | Ahmedabad, Gujarat |
| Employment Type | Full-Time |
| Qualification | B.Pharm / M.Pharm / B.Sc / M.Sc / Engineering |
| Experience | 0 to 6 Years |
| Salary | ₹2.0 LPA – ₹8.0 LPA |
| Application Mode |
Job Location & Employment Type
The job location is Ahmedabad, Gujarat, and all roles are full-time, on-site positions. Candidates must be willing to work in a manufacturing environment and adhere to company policies and compliance standards.
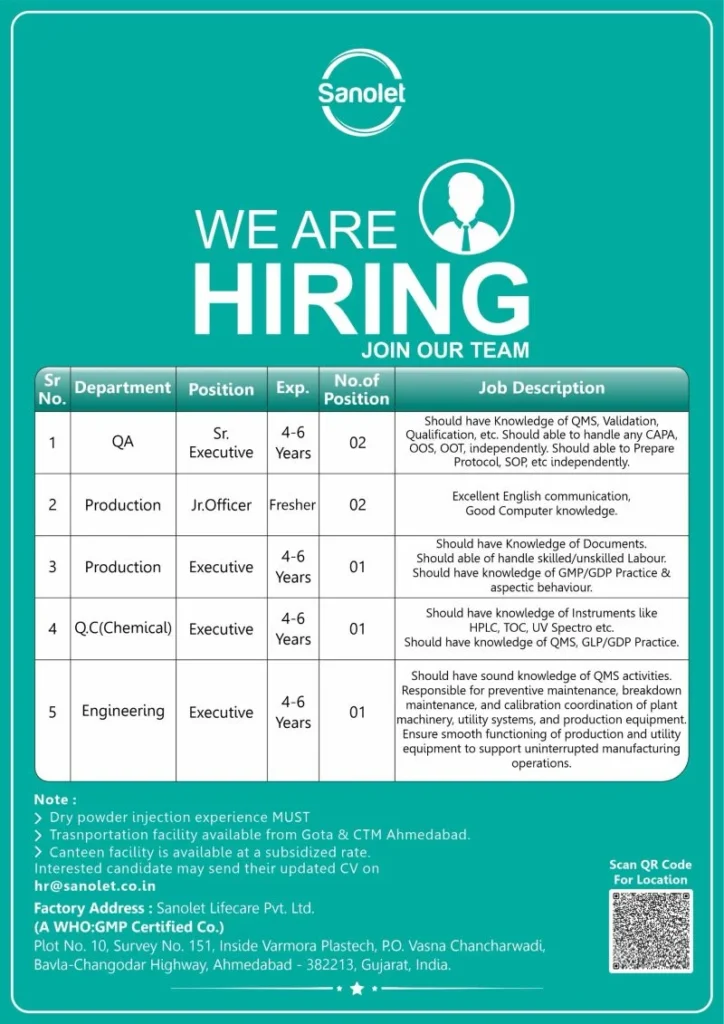
Open Positions / Department Details
Sanolet Lifecare is hiring across multiple departments with the following roles:
- Quality Assurance – Senior Executive
- Production – Junior Officer (Freshers)
- Production – Executive
- Quality Control (Chemical) – Executive
- Engineering – Executive
Each position offers distinct responsibilities and career growth opportunities within the pharmaceutical manufacturing domain.
Key Roles & Responsibilities
Quality Assurance (QA – Sr. Executive)
- Manage Quality Management Systems including documentation and compliance
- Handle validation and qualification activities
- Investigate and manage deviations such as CAPA, OOS, and OOT
- Prepare and review SOPs, protocols, and reports
- Ensure adherence to regulatory and quality standards
Production – Junior Officer (Fresher)
- Maintain batch manufacturing records and documentation
- Follow GMP guidelines during production processes
- Assist in daily manufacturing activities
- Coordinate with team members for smooth workflow
- Ensure proper record-keeping and reporting
Production – Executive
- Supervise production teams including operators and workers
- Ensure compliance with GMP, GDP, and aseptic practices
- Operate and monitor pharmaceutical instruments such as HPLC, TOC, and UV spectrophotometers
- Maintain production efficiency and quality standards
- Handle troubleshooting during manufacturing processes
Quality Control (QC – Chemical – Executive)
- Perform analytical testing and chemical analysis
- Ensure laboratory compliance with GLP and GDP standards
- Maintain proper documentation and records
- Operate and calibrate laboratory instruments
- Participate in method validation and quality checks
Engineering – Executive
- Conduct preventive and breakdown maintenance of equipment
- Manage calibration and utilities
- Ensure uninterrupted production operations
- Maintain engineering documentation
- Support production and quality teams for technical issues
Eligibility Criteria
Education:
- B.Pharm
- M.Pharm
- B.Sc
- M.Sc
- Engineering (relevant disciplines)
Experience:
- Freshers are eligible for junior-level roles
- 4 to 6 years of experience required for senior and executive roles
Skills Required:
Technical Skills:
- Knowledge of GMP, GLP, GDP, and QMS
- Understanding of pharmaceutical manufacturing processes
- Experience with laboratory and production instruments
- Documentation and compliance handling
Soft Skills:
- Good communication skills
- Team coordination and collaboration
- Attention to detail
- Problem-solving ability
- Basic computer proficiency
Salary & Benefits
- Salary ranges from ₹2.0 LPA to ₹8.0 LPA depending on experience and role
- Subsidized canteen facility
- Transportation facility from Gota and CTM areas in Ahmedabad
- Opportunity to work in a WHO-GMP certified organization
- Exposure to regulated pharmaceutical manufacturing processes
- Career growth and skill development opportunities
Selection Process
The selection process may include the following stages:
- Initial screening of applications
- Technical interview with department heads
- HR interview for final selection
- Final offer and onboarding
Candidates are advised to prepare for both technical and general interview questions related to their field.
How to Apply
Interested candidates can apply by sending their updated resume to the official email ID:
Make sure your CV is updated with relevant qualifications, experience, and contact details before applying.
Why Apply for This Pharma Job?
This opportunity at Sanolet Lifecare Pvt. Ltd. is highly beneficial for both freshers and experienced candidates aiming to establish a strong career in the pharmaceutical industry. The company provides exposure to advanced manufacturing practices, regulatory compliance, and quality systems. Fresh graduates can gain hands-on experience, while experienced professionals can take on leadership roles and enhance their expertise.
Working in a WHO-GMP certified organization also strengthens your professional profile and opens doors to future opportunities in reputed pharma companies. Additionally, the availability of facilities like transportation and subsidized meals adds convenience for employees.
Important Note / Disclaimer
PharmaJobHub.in is an independent job information platform. We do not provide any recruitment guarantee and are not affiliated with any company. Candidates are advised to verify details from official sources before applying.
Final Call-to-Action
Candidates who meet the eligibility criteria and are interested in building a career in pharmaceutical manufacturing are encouraged to apply as soon as possible. Early applications increase the chances of selection, especially for limited vacancies.


