RL Fine Chem Hiring for QC QA EHS Jobs in Hosur for experienced pharmaceutical API professionals.
The pharmaceutical API industry continues to create strong career opportunities for professionals specializing in quality control, quality assurance, and environmental safety management. Experienced candidates looking to work with a regulatory-driven pharmaceutical manufacturing organization now have a promising opportunity.
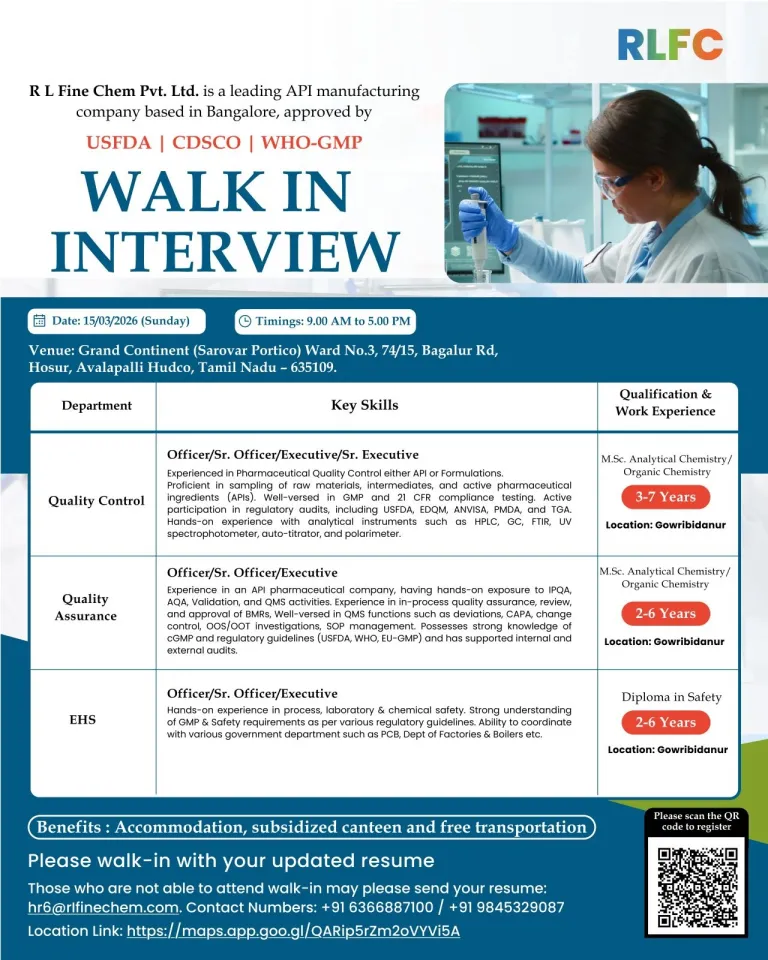
RL Fine Chem Pvt. Ltd. has announced a walk-in recruitment drive for multiple roles in the Quality Control (QC), Quality Assurance (QA), and Environment, Health & Safety (EHS) departments. The company is inviting qualified candidates with relevant experience in the pharmaceutical API sector to attend the interview scheduled in Hosur, Tamil Nadu.
This hiring initiative is particularly suitable for professionals with analytical chemistry backgrounds, regulatory compliance knowledge, and safety management expertise who want to advance their careers in a globally compliant API manufacturing environment. RL Fine Chem Hiring for QC QA EHS Jobs
Company Overview
RL Fine Chem Pvt. Ltd. is a pharmaceutical manufacturing company specializing in Active Pharmaceutical Ingredients (APIs). The company operates modern manufacturing facilities that follow strict global quality standards and regulatory requirements.
The organization is recognized for maintaining compliance with international regulatory authorities such as USFDA, CDSCO, and WHO-GMP. With a strong focus on quality systems, regulatory compliance, and advanced manufacturing processes, RL Fine Chem provides professionals with opportunities to work in globally regulated pharmaceutical environments and build specialized expertise. RL Fine Chem Hiring for QC QA EHS Jobs
Job Overview
| Job Title | Quality Control, Quality Assurance & EHS Jobs |
|---|---|
| Company | RL Fine Chem Pvt. Ltd. |
| Job Location | Gowribidanur |
| Interview Location | Hosur, Tamil Nadu |
| Employment Type | Full-Time |
| Experience Required | 2 – 7 Years |
| Qualification | M.Sc (Analytical Chemistry / Organic Chemistry), Diploma in Safety |
| Interview Mode | Walk-In Interview |
| Interview Date | 15 March 2026 |
| Interview Timing | 9:00 AM – 5:00 PM |
| Industry | Pharmaceutical / API Manufacturing |
Job Location & Employment Type
Selected candidates will be placed at the company’s manufacturing facility located in Gowribidanur. These roles involve working in a regulated pharmaceutical manufacturing environment where quality, compliance, and operational safety are critical.
All positions are full-time roles within the pharmaceutical API sector, offering opportunities for technical skill development and long-term career growth. RL Fine Chem Hiring for QC QA EHS Jobs
Open Positions / Department Details
RL Fine Chem is hiring experienced professionals for the following departments:
Quality Control (QC)
The Quality Control department is responsible for analytical testing of pharmaceutical products and raw materials to ensure compliance with global quality standards.
Quality Assurance (QA)
The QA department focuses on maintaining quality systems, documentation, compliance management, and regulatory readiness across the manufacturing facility.
EHS – Environment, Health & Safety
The EHS department ensures that manufacturing operations meet environmental regulations and workplace safety standards while protecting employees and the surrounding environment. RL Fine Chem Hiring for QC QA EHS Jobs
Key Roles & Responsibilities
Quality Control (Officer / Sr. Officer / Executive / Sr. Executive)
- Performing analytical testing of APIs or pharmaceutical formulations
- Conducting analysis of raw materials, intermediates, and finished products
- Operating laboratory instruments such as HPLC, GC, FTIR, UV spectrophotometer, and auto-titrator
- Ensuring laboratory operations follow GMP and regulatory requirements including 21 CFR guidelines
- Preparing and maintaining laboratory documentation and analytical reports
- Supporting validation activities and method development processes
- Participating in regulatory inspections and audits conducted by agencies such as USFDA, EDQM, ANVISA, PMDA, and DCGI
Quality Assurance (Officer / Sr. Officer / Executive)
- Managing quality systems including QMS documentation and compliance procedures
- Handling activities related to IPQA, AQA, and process validation
- Investigating deviations and implementing corrective and preventive actions (CAPA)
- Managing change control processes and quality investigations including OOS and OOT events
- Supporting internal audits and regulatory inspections
- Ensuring compliance with global regulatory guidelines such as USFDA, WHO-GMP, and EU-GMP
EHS (Officer / Sr. Officer / Executive)
- Implementing environmental health and safety programs across manufacturing and laboratory operations
- Monitoring safety practices and ensuring compliance with regulatory requirements
- Coordinating with regulatory authorities such as the Pollution Control Board and the Department of Factories & Boilers
- Conducting workplace safety assessments and implementing risk control procedures
- Promoting employee safety awareness and ensuring adherence to industrial safety standards
Eligibility Criteria
Education
Candidates should possess the following educational qualifications depending on the role:
- M.Sc in Analytical Chemistry or Organic Chemistry for QC and QA roles
- Diploma in Safety for EHS roles
Candidates with strong academic backgrounds and pharmaceutical industry exposure will be preferred.
Experience
- Quality Control: 3 – 7 years of relevant pharmaceutical experience
- Quality Assurance: 2 – 6 years of experience in API manufacturing
- EHS: 2 – 6 years of experience in industrial safety or pharmaceutical manufacturing environments
Experience in regulated pharmaceutical facilities is considered an advantage.
Skills Required
- Strong understanding of GMP and regulatory compliance requirements
- Experience working with analytical instruments such as HPLC and GC
- Knowledge of pharmaceutical quality systems and documentation practices
- Investigation and problem-solving skills related to deviations and quality issues
- Regulatory audit readiness and compliance knowledge
- Safety management and risk assessment skills for EHS roles
- Good communication and documentation abilities
RL Fine Chem Hiring for QC QA EHS Jobs
Salary & Benefits
RL Fine Chem offers competitive salary packages depending on the candidate’s role, qualifications, and industry experience.
Estimated Salary Range
₹3.5 LPA – ₹8 LPA
Employee Benefits May Include
- Accommodation assistance for employees
- Subsidized canteen facilities
- Transportation support for employees
- Opportunity to work in a USFDA-approved pharmaceutical manufacturing environment
- Exposure to global regulatory standards and audits
- Professional growth within a regulated pharmaceutical organization
RL Fine Chem Hiring for QC QA EHS Jobs
Selection Process
The recruitment process will be conducted through a walk-in interview. Candidates attending the interview may undergo the following evaluation steps: RL Fine Chem Hiring for QC QA EHS Jobs
- Candidate registration and initial screening
- HR interaction to evaluate experience and eligibility
- Technical interview with department experts
- Document verification and final selection
Shortlisted candidates may receive further instructions regarding employment formalities. RL Fine Chem Hiring for QC QA EHS Jobs
How to Apply
Interested candidates can attend the walk-in interview at the following venue with the required documents.
Interview Venue
Grand Continent (Sarovar Portico)
Ward No. 3, 74/15, Bagalur Rd
Hosur, Avalapalli Hudco
Tamil Nadu – 635109, India
Candidates should bring the following documents:
- Updated resume or CV
- Educational qualification certificates
- Experience certificates
- Government ID proof
Candidates who are unable to attend the walk-in interview may also send their resume via email.
Email: hr6@rlfinechem.com
Contact Numbers
+91 6366887100
+91 9845329087
Applicants are advised to reach the venue early to complete the registration process and avoid delays. RL Fine Chem Hiring for QC QA EHS Jobs
Important Dates
Walk-In Interview Date: 15 March 2026
Interview Time: 9:00 AM – 5:00 PM
Candidates planning to attend should arrive within the interview timing window with all required documents. RL Fine Chem Hiring for QC QA EHS Jobs
Why Apply for This Pharma Job?
Working in the pharmaceutical API industry offers professionals exposure to regulated manufacturing processes and international quality standards. RL Fine Chem provides opportunities for professionals to work in an environment that emphasizes regulatory compliance, safety management, and operational excellence. RL Fine Chem Hiring for QC QA EHS Jobs
Key advantages of this opportunity include:
- Multiple openings for experienced pharmaceutical professionals
- Exposure to global regulatory inspections and compliance systems
- Opportunity to work in a USFDA-approved manufacturing facility
- Access to professional development in quality and safety management
- Additional employee benefits such as accommodation and transportation support
Professionals seeking stable career growth in pharmaceutical quality, regulatory compliance, or safety management can benefit from this opportunity. RL Fine Chem Hiring for QC QA EHS Jobs
Important Note / Disclaimer
Pharmajobhub.in is an independent pharmaceutical job information platform. We do not participate in recruitment processes or charge any fees for job applications.
Our goal is to share verified pharmaceutical job opportunities for job seekers across India. Candidates should verify job details independently and communicate directly with the hiring company for recruitment-related queries. Pharmajobhub.in is not affiliated with RL Fine Chem Pvt. Ltd.
Final Call-to-Action
Eligible candidates interested in building a career in pharmaceutical quality control, quality assurance, or safety management should consider attending this walk-in interview. Preparing the required documents and arriving on time can help improve the chances of selection.
Stay connected with Pharmajobhub.in for the latest pharmaceutical job updates in production, quality, research, regulatory affairs, and healthcare industries. RL Fine Chem Hiring for QC QA EHS Jobs


