Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
Company: Gracure Pharmaceuticals
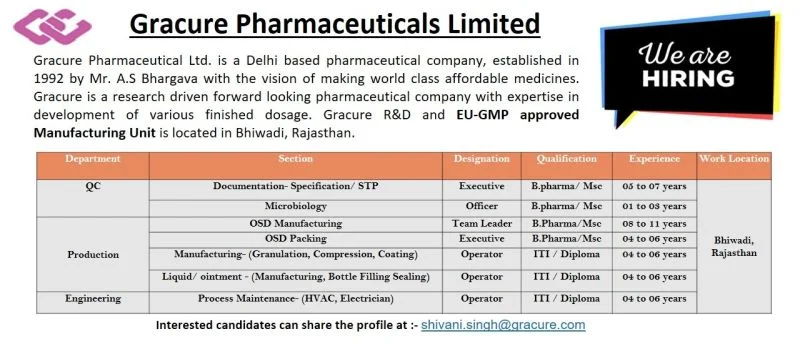
Job Location: Bhiwadi, Rajasthan
Industry: Pharmaceutical Manufacturing
Job Type: Full-Time
Experience Required: 1 – 11 Years
Qualifications: B.Pharm, M.Sc, ITI, Diploma
Job Status: Verified Job
Application Mode: Email Application
Company Overview
Gracure Pharmaceuticals Limited, established in 1992, is a Delhi-based, research-driven pharmaceutical company with a strong presence in finished dosage formulations. The company operates an EU-GMP approved manufacturing facility in Bhiwadi, Rajasthan, and is known for producing high-quality, affordable medicines while maintaining strict regulatory compliance. Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
As part of its expansion, Gracure Pharmaceuticals is inviting applications from experienced and skilled professionals for Quality Control (QC), Production, and Engineering roles. Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
Open Positions & Key Responsibilities
Quality Control (QC)
QC Executive – Documentation (Specification / STP)
Responsibilities:
- Preparation, review, and maintenance of specifications and Standard Testing Procedures (STP)
- Ensure documentation compliance with regulatory and quality standards
QC Officer – Microbiology
Responsibilities:
- Perform microbial testing and environmental monitoring
- Ensure sterility assurance and microbiological compliance
- Maintain microbiology-related documentation
Production Department
OSD Manufacturing Team Leader / Executive
Responsibilities:
- Oversee Oral Solid Dosage (OSD) manufacturing operations
- Manage granulation, compression, and coating processes
- Achieve production targets while ensuring GMP compliance
OSD Packing Team Leader
Responsibilities:
- Supervise OSD packing activities
- Ensure batch accuracy, documentation, and timely dispatch
- Maintain compliance with GMP and safety guidelines
Production Operator – Liquid / Ointment
Responsibilities:
- Operate equipment for liquid and ointment manufacturing
- Handle bottle filling, sealing, and related production activities
- Ensure adherence to SOPs and safety standards
Engineering Department
Engineering Operator – Process Maintenance (HVAC / Electrician)
Responsibilities:
- Maintain HVAC systems, electrical equipment, and utilities
- Support uninterrupted production through preventive maintenance
- Ensure compliance with safety and engineering SOPs
Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
Eligibility Criteria & Experience
- QC Executive: B.Pharm / M.Sc with 5–7 years experience
- QC Officer (Microbiology): B.Pharm / M.Sc with 1–3 years experience
- OSD Manufacturing Team Leader: B.Pharm / M.Sc with 8–11 years experience
- OSD Manufacturing Executive: B.Pharm / M.Sc with 4–6 years experience
- OSD Packing Team Leader: B.Pharm / M.Sc with 4–6 years experience
- Production Operator: ITI / Diploma with 4–6 years experience
- Engineering Operator: ITI / Diploma with 4–6 years experience
Experience in EU-GMP or other regulated pharmaceutical manufacturing environments is highly preferred.
Salary & Benefits
Gracure Pharmaceuticals offers industry-aligned compensation packages:
- Executives / Team Leaders: ₹6 – 12 LPA
- Officers: ₹4 – 8 LPA
- Operators: ₹3 – 6 LPA
Benefits may include:
- Provident Fund (PF)
- Health Insurance
- Performance-based incentives
- Long-term career growth opportunities
Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
How to Apply
Interested candidates should email their updated resume to:
Email ID: shivani.singh@gracure.com
Work Location: Bhiwadi, Rajasthan (EU-GMP approved facility)
This is a great opportunity to build or advance your career with a reputed pharmaceutical manufacturing organization known for quality and compliance. Gracure Pharmaceuticals Hiring for QC Production Engineering Roles
For verified pharma job updates, visit PharmaJobHub.in – India’s trusted pharma career portal.
Note: This job information is sourced from publicly available official sources. We share it for reference purposes only and do not claim ownership of the job posting.


